

| Archive Blog Cast Forum RSS Books! Poll Results About Search Fan Art Podcast More Stuff Random |
|
Classic comic reruns every day
|
1 {photo of Canterbury College in Christchurch, where Ernest Rutherford studied}
1 Caption: Probing the atom
|
First (1) | Previous (3213) | Next (3215) || Latest Rerun (2873) |
Latest New (5380) First 5 | Previous 5 | Next 5 | Latest 5 Annotations theme: First | Previous | Next | Latest || First 5 | Previous 5 | Next 5 | Latest 5 This strip's permanent URL: http://www.irregularwebcomic.net/3214.html
Annotations off: turn on
Annotations on: turn off
|
 A rock, being pushed. |
What is an electric charge, exactly? That's a difficult question to answer on the most fundamental level, but functionally an electric charge is something that feels a force when in the presence of another electric charge[1]. It might be pushed away or pulled towards that other charge. This property, plus the fact that electric charges can be made to move around, results in all of the various phenomena we associate with electricity.
So, electric charge exists. How does it relate to matter and, specifically, how is it related to atoms? The first attempt to answer this was by British scientist J. J. Thomson, the man who discovered the electron in 1897. He realised that electric charge was carried by particles that were contained within atoms. Atoms were usually electrically neutral, he said. That is, they contained the same amount of positive electric charge as negative electric charge. When you rub two suitable materials together, electrons (which we discussed previously) get pulled out of the atoms of one material and deposited on to the other material. This explanation by itself is enough to explain the known properties of electricity at the time.
 Plum pudding. |
Thomson's plum pudding model of the atom lasted for five years. One of his own former students directed the experiment that disproved it in 1909. Ernest Rutherford supervised Hans Geiger (inventor of the Geiger counter) and Ernest Marsden in the experiment, in which they fired positively charged helium atoms (i.e. with the electrons stripped off) at a thin sheet of gold foil. It was known at the time that the helium atoms passed through the gold foil, indicating that atoms were diffuse in some sense. A sheet of atoms was not like a brick wall that stopped anything passing through; it was more like a sieve, which let objects as small as other atoms pass straight through. A bit like bullets being fired at a wall made of plum puddings, in fact.
 Gold foil. (actually, aluminium foil, coloured gold...) |
To their surprise, they found that there was almost no deflection at all. Pretty much all of the charged helium atoms passed through completely unscathed, with a deflection angle of zero degrees. A very few helium atoms were deflected by small angles.
Just for completeness, they swivelled their detector all the way around the gold foil, and then a most astonishing thing happened. Some of the charged helium atoms had been deflected by really large angles, 90 degrees or more. What's more, some of them had bounced off the foil, almost straight back where they came from! This was completely unexpected. In Rutherford's own words:
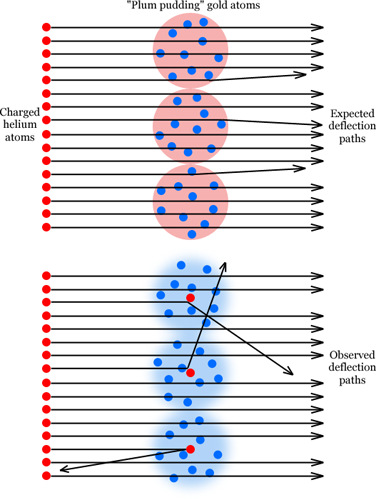 Top: Charged helium atoms fired at gold foil made of plum pudding atoms, with expected deflection paths. Bottom: Observed results of the experiment. Positive electric charges are red, negative charges are blue. |
It was quite the most incredible event that has ever happened to me in my life. It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.
What was going on here? It took Rutherford and his colleagues two years to formulate and publish their explanation, in 1911. The only way to explain what they had observed was to conclude that the positively charged part of the gold atoms was extremely small, in fact much smaller than the size of the atoms themselves. Doing the calculations, the positive charge in a gold atom had to be about 1/4000 of the size of the atom overall. This way, nearly all of the charged helium atoms hitting the gold foil would simply pass through empty space, but occasionally one would come close to hitting this nucleus and be deflected by a substantial angle, and sometimes one would hit it almost dead on and be bounced straight back to where it came from.
Rutherford's model of the atom consisted of a tiny, positively charged nucleus, which contained all of the positive charge of the atom. The electrons had to be further away, not embedded within the nucleus like Thomson's plum pudding. But remember that positive charges (like the nucleus) and negative charges (like the electrons) are attracted to one another. How could they be held apart? To explain this, Rutherford took inspiration from another physical system in which objects that are mutually attracted to one another are held at a great distance in a stable arrangement: planets in orbit around the sun. In Rutherford's model of the atom, the electrons were like planets, orbiting at a relatively large distance around a small nucleus, and held in this arrangement by the attraction of their electric charges.
This result implied that the electrons were an important part of what makes up an atom, not merely unnecessary adornments like plums in a pudding. In Thomson's model, the pudding was the atom, and (to mix metaphors) the electrons were like fleas that just hitched along for the ride. In Rutherford's model, the electrons were the outer layers of the atom. If you stripped the electrons away, you wouldn't have a plumless pudding atom left over, you'd have something much, much smaller than an atom. The distinction may sound subtle, but Rutherford's result was the first really strong hint that atoms were in fact not indivisible balls of matter (that attracted the mysterious electrons). Atoms were made of smaller particles.
This was a closer model to reality than the plum pudding atom but, as it turned out, Rutherford's model lasted an even shorter time than Thomson's five years. The problem with Rutherford's model was that it required the charged electrons to move in circular (or elliptical) orbits around the nucleus. In 1897, Joseph Larmor had shown, using Maxwell's equations of electromagnetism (which I've discussed previously - you may want to refresh your memory), that an electric charge moving in a circle will emit electromagnetic radiation (i.e. light, or radio waves, or x-rays, etc.) So the electrons in Rutherford's model should be constantly emitting radiation. And when they emit radiation, they lose energy, which means they can't stay in the same orbit - they should spiral in towards the nucleus[3].
In other words, if atoms were really like Rutherford's model, then they should all spontaneously collapse in a flash of radiation. They obviously don't do this, so something was wrong.
 Visible spectrum of hydrogen. Adapted from public domain image from Wikimedia Commons. |
Rydberg made his formula work for the non-visible lines of the hydrogen spectrum as well - those in the ultraviolet and infrared parts of the spectrum. His formula looks like this:
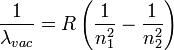
 Some red, hot objects. |
Rydberg published his result in 1888. It was a triumph of experimental physics. The only trouble was that nobody had any idea why it worked.
In 1912, a year after Rutherford published his model of the atom, the Danish physicist Neils Bohr joined his research team in Manchester. Bohr was disturbed by both the theoretical problem in Rutherford's atomic model, and the lack of any physical explanation for Rydberg's formula. He addressed both of these in a single brilliant insight, by using a third piece of recent physics.
In order to explain the observed spectrum of light emitted by a hot object (a red hot piece of iron, the sun, a white hot material, etc.), Max Planck had formulated a theory in 1901, in which electromagnetic radiation is emitted not continuously, but is quantised into separate, discrete "packets" of energy. The details are too much to go into now, but his main result was that when an electric charge oscillates in a hot object, it doesn't lose energy continuously, but rather in discrete jumps. Furthermore the jumps in energy are related to the wavelength of the light that ends up being emitted, by the simple formula:
E = hν
 Jumping between energy levels. |
Bohr realised that Planck's treatment of hot oscillating charges could also be applied to the movement of electrons in an atom. Bohr proposed that electrons orbiting a nucleus could also not lose energy continuousy (as predicted by Maxwell's equations), but only in discrete jumps, with energy related to the wavelength of light by Planck's formula. Certain fixed orbits of the electrons were therefore stable over long periods of time, and the electrons could only "jump" between the orbits by gaining or losing a fixed amount of energy. The energies of the orbits were such that the angular momentum of electrons circling the nucleus were whole number multiples of Planck's constant divided by 2π (that factor comes in because we're talking about angular momentum instead of energy):
(angular momentum) L = n / 2π; where n is a whole number.
By making these assumptions, Bohr showed that the radiation spectrum emitted by a hydrogen atom could be explained as the electrons jumping between orbits of differing energies. This matched Rydberg's formula, with n1 and n2 being precisely equal to the whole number multiples n of the two different orbits which the electron jumps from, and jumps to. Furthermore, this model allowed Bohr to calculate the value that the Rydberg constant R shoud have, in terms of the Planck constant, the mass of the electron, the charge of the electron, the speed of light, and the permittivity of free space constant from Maxwell's equations. And when Bohr plugged in all these numbers, he got the same value for the Rydberg constant that Rydberg had measured experimentally. How good is that?
 Humboldt University, Berlin. Where Max Planck taught physics. |
Bohr's model has since been superseded, but it still survives as the first mathematically rigorous and useful approximation taught to physics students today. (Similar to how students are first taught Newton's laws of motion, rather than thrown straight into Einstein's special and general relativity.) Furthermore, in historical context, Bohr's model was a crucial stepping stone to the newly developing theory of quantum mechanics that would change the world in the next few decades.
In 1913, less than a hundred years ago, we had our first glimpse of understanding of how atoms really work.
Addendum: I mentioned in an earlier annotation that electricity is the answer to two puzzling historical questions about the properties of atoms and elements. We're getting closer, but we still need a little bit more work to answer those questions:
[2] Ha, currants. Get it?
[3] In the same way that satellites that lose energy by dragging through the topmost parts of Earth's atmosphere eventually spiral down and crash into Earth. (In an even closer analogy, satellites also lose energy by radiating gravitational waves—"waves of gravity"—which are somewhat similar to electromagnetic radiation being "waves of electric field". However, the loss of energy from a satellite by gravitational radiation is too small to be measured, and vastly overwhelmed by the loss due to upper atmospheric friction.)
|
LEGO® is a registered trademark of the LEGO Group of companies,
which does not sponsor, authorise, or endorse this site. This material is presented in accordance with the LEGO® Fair Play Guidelines. |